Melanotan II Research: Quality Checks & Handling Guide
The fastest way to lose clarity in peptide research is to let your workflow drift. A vial prepared one way by one person, a different way by another, and then the lab compares outcomes as if the setup was identical. It was not. In receptor-driven models, small preparation differences can masquerade as real changes, making troubleshooting slow and frustrating.
That is why Melanotan II research should start with the fundamentals. Tight intake documentation. Storage habits that stay consistent. Preparation math that does not change depending on who is doing the prep. When those are locked in, your experiments become easier to interpret and easier to reproduce.
If you are sourcing this compound, start with Melanotan II Peptide and treat it like a controlled research input from the moment it arrives.
What Melanotan II Means in a Research Workflow
In research discussions, Melanotan 2 is commonly referenced in melanocortin-pathway and receptor signaling models. The exact protocol varies by team, but the workflow requirement stays the same: stable inputs produce interpretable outcomes.
With MT2 peptide, your team should be able to answer these questions without guessing:
- Which lot did we use?
- Where is the COA for that lot?
- What concentration did we prepare and when?
- How was the vial stored and accessed between runs?
If those answers are clear, troubleshooting stays quick and comparisons remain meaningful. For consistent naming and centralized sourcing, use Peptides as your inventory reference.
Why Labs See Inconsistent Outcomes with Melanotan II
Most inconsistency comes from routine drift rather than a dramatic “bad vial” scenario:
- A different reconstitution volume is used by a different researcher.
- A label is vague, so someone assumes the concentration.
- The vial is accessed more often during a busy week, increasing temperature cycling.
- A new lot arrives but is not recorded in the experiment notes.
Then the lab expects replicates to line up, but they cannot, because the input was not truly consistent. If you tighten intake and prep discipline for Melanotan II peptide, these problems drop fast.
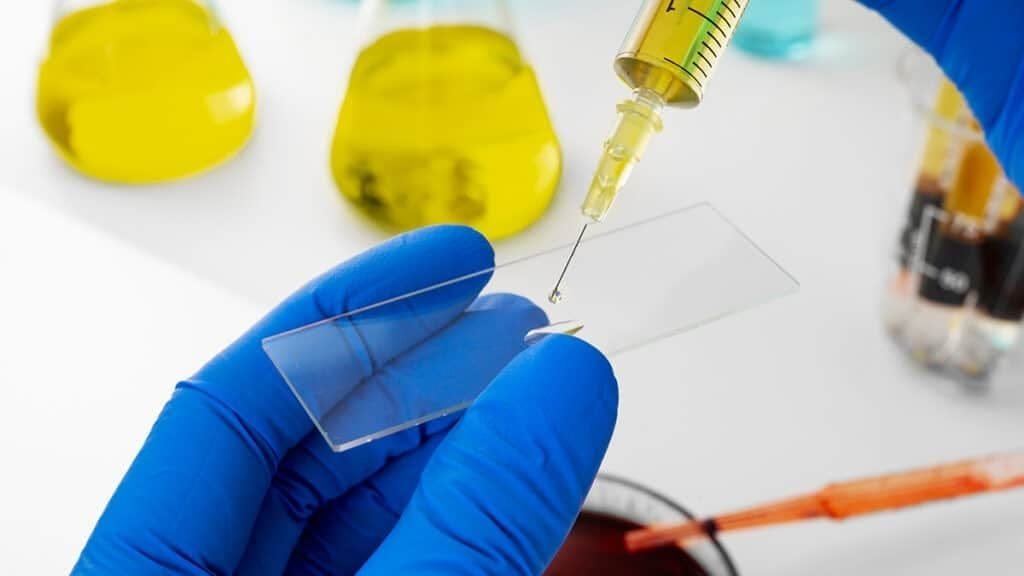
COA Review: The Intake Habit That Protects Your Study
A Certificate of Analysis is part of your experiment record. Before you prepare this melanocortin agonist, confirm that the COA matches the vial and contains the key details your lab needs for traceability.
Lot Number Match
Confirm the lot or batch number on the vial matches the COA exactly. If it does not, pause and resolve it before prep. Without lot traceability, long-term comparisons become guesswork.
Stated Analytical Method
Purity should be tied to a stated method. Many peptide COAs reference HPLC profiling. Your goal is not to overanalyze the chemistry. Your goal is to confirm the method is stated clearly enough to log consistently.
Lot-Specific Documentation
A COA should look lot-specific rather than generic. Clear documentation makes it easier to confirm whether a change in outcomes aligns with a lot change.
Keep this same discipline across your inventory, whether you are logging Melanotan II peptide, PT-141 Peptide, or Epitalon 50mg.
Purity in Practical Terms: What Quality Really Means
Purity matters because impurities and degradation products can introduce background noise that looks like inconsistent receptor behavior. The tricky part is that this noise can look like real effects, leading teams down the wrong path.
With Melanotan II peptide, quality comes down to two essentials:
- Verification of what arrived
- Protection of what arrived through consistent handling
Even clean material can become inconsistent if it is repeatedly warmed and cooled, left exposed during prep, or prepared at different concentrations across researchers. Think of purity verification as baseline confidence and your SOP as what protects that baseline.
Storage and Handling: Small Habits That Protect Stability
Most peptide stability issues are caused by predictable habits: bench time, exposure, and temperature cycling. Here is how to address them.
Keep Bench Time Short
Open the vial only when needed, work efficiently, seal it, and return it to controlled storage quickly. Avoid leaving it out while you do unrelated tasks. This is one of the easiest ways to keep Melanotan II peptide stable across multiple runs.
Reduce Repeated Warm-Cold Cycles
Repeated temperature swings increase gradual degradation risk over time. If repeated use is expected, structure your workflow to reduce how often the same container is warmed, opened, and returned. Many labs prepare a controlled stock under one documented routine, then work from smaller portions. The method should match your internal standards; what matters is consistency.
Standardize Storage Behavior Across the Team
Shared inventory needs shared habits. If one person handles the vial quickly and another leaves it out longer, the compound experiences different conditions. Standardized access behavior keeps Melanotan 2 peptide consistent over time.
Preparation and Concentration: Keep It Consistent
Most labs run into concentration drift because documentation is incomplete, not because the math is difficult.
For Melanotan II peptide, choose one standard reconstitution volume for the project and stick to it. Then record volume and concentration together in the same format every time.
A clean prep record includes:
- Reconstitution volume
- Final concentration
- Prep date
- Lot number
- Initials of preparer
If your team wants one shared conversion standard, use Peptide Calculator so everyone calculates the same way and logs results consistently.
A Repeatable Workflow Your Team Can Follow
Step 1: Receive and Log
Log arrival date, product name, and lot number on the day it arrives. Store the COA with the lot record. Use the product page as your naming reference: Melanotan II Peptide.
Step 2: Verify Before First Use
Match the COA lot number to the vial label. Confirm the analytical method is stated and the COA looks lot-specific.
Step 3: Store Immediately and Consistently
Move the vial into controlled storage quickly. Keep bench time short. Keep access habits consistent across team members.
Step 4: Prepare Using One Lab Standard
Pick a standard reconstitution volume for Melanotan II peptide and do not improvise mid-study. If another project needs a different concentration, treat it as a separate prep batch and label it clearly.
Step 5: Track Usage Across Runs
Record lot number and preparation batch details in each run’s notes. If outcomes drift, you can quickly check whether the drift aligns with a lot change, a prep change, or a storage access pattern.
Avoiding Mix-Ups with PT-141
Melanotan II and PT-141 are often discussed in similar research circles, but they are different compounds and should not share assumptions in labeling, prep standards, or documentation. If your lab stocks both, keep records clearly separated and label prepared stocks precisely using product names.
Use Peptides as the centralized inventory list so your team uses consistent names and references.

Common Mistakes That Quietly Ruin Comparability
If Melanotan II peptide outcomes start looking inconsistent, check these first:
- Did the reconstitution volume change between runs?
- Did the lot number change without being recorded?
- Was the vial accessed more often than usual, increasing temperature cycling?
- Were concentrations logged in inconsistent units or formats?
- Did different researchers handle the vial with different bench-time habits?
Fixing intake and prep discipline is usually faster than rewriting the protocol.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Use one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference helps keep conversions consistent.
Why does lot tracking matter so much in Melanotan II research?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly check whether the shift aligns with a lot change rather than questioning your model.
Where should new team members look to understand what we stock?
Use Peptides as the centralized inventory list so naming and sourcing stay consistent across the lab.
Research Use Disclaimer: Melanotan II (MT2) peptide is sold strictly for research and laboratory use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should consult all applicable institutional guidelines and regulations before use.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Use one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference helps keep conversions consistent.
Why does lot tracking matter so much in Melanotan II research?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly check whether the shift aligns with a lot change rather than questioning your model.
Where should new team members look to understand what we stock?
Use the Peptides catalog as the centralized inventory list so naming and sourcing stay consistent across the lab.
NAD+ Peptide Research: COA, Storage & Reconstitution Guide
Have you ever watched a promising NAD+ experiment produce beautiful data in week one, only to see subtle inconsistencies creep in by week three? You’re not alone. In most cases, the assay isn’t failing—your inputs are drifting. NAD+ peptide research is particularly vulnerable to this because studies often span months, involve multiple researchers, and require careful coordination across teams.
The good news? Most of this variability disappears when you lock down three fundamentals: verifying what you received, storing it correctly, and preparing it the same way every single time. This guide walks you through exactly how to do that.
If you’re building your research inventory, you can start with NAD+ 500MG and implement these workflow standards from day one.
Understanding NAD+ in Research Contexts
NAD+ sits at the center of cellular energy metabolism and redox biology, making it a cornerstone compound for labs studying foundational cellular processes. Its broad applicability across experimental models is both a strength and a challenge—precisely because so many teams use it, handling discipline can slip.
When you’re working with NAD+ peptide, you want every vial to be a documented, traceable asset in your experimental record. That means lot numbers logged, COAs attached, storage conditions standardized, and preparation math identical across every run. No exceptions.
For labs managing multiple compounds, centralizing your inventory through the Peptides catalog keeps naming and documentation consistent—no more patchwork records or guesswork.
Why NAD+ Results Drift (And It’s Not Your Assay)
Let’s be honest about how drift actually happens in busy labs. It’s never dramatic—it’s cumulative.
One researcher reconstitutes with 2 mL, another uses 2.5 mL, and neither writes it down clearly. A vial gets pulled from the freezer repeatedly during a hectic week, warming and cycling more than anyone realizes. Prep notes read “made stock” instead of “reconstituted 10 mg in 2 mL bacteriostatic water = 5 mg/mL.” Then a new lot arrives, but the lot number never makes it into the experiment file.
Suddenly you’re comparing datasets that were never truly comparable—and worse, you don’t realize it until you’ve burned days troubleshooting biology that was never the problem.
The solution isn’t complicated. It’s just disciplined. A tighter intake and prep routine for NAD+ peptide keeps your compound predictable across your entire timeline.
COA Verification: Your Five-Minute Insurance Policy
Think of your Certificate of Analysis as part of your experimental record, not bureaucratic paperwork. Before any NAD+ peptide enters your workflow, your COA needs to answer one critical question: can you defend what this vial is, and can you tie it to a specific, traceable lot?
What to verify before the vial touches your bench
Check the lot number first. It must match the vial label exactly. If it doesn’t match, stop. Lot traceability is the bedrock of meaningful experimental comparisons—without it, you’re building on sand.
Confirm the analytical method. Purity values are meaningless without context. The COA should clearly state how verification was performed—whether that’s HPLC profiling, mass spectrometry, or another validated approach. You’re not trying to become an analytical chemist; you just need documented, defensible methodology.
Ensure it’s lot-specific documentation. Generic-looking COAs create generic records, and generic records create troubleshooting nightmares down the road. Your documentation should clearly tie to the specific lot in your hand.
This verification step becomes even more critical when you’re running NAD+ peptide across multiple timepoints. When results shift, you need to distinguish between a lot change, a storage change, and a prep change—and your COA is the first place you look.
Purity in Practice: Beyond the Numbers
Labs sometimes treat purity as a specification to check and file away. In reality, purity is a reproducibility issue. Impurities introduce background noise, and even small shifts can manifest as variability in sensitive readouts.
But purity alone isn’t enough. A high-quality vial handled carelessly becomes an inconsistent input just as surely as a questionable one. With NAD+ peptide, purity verification and handling discipline work hand-in-hand:
Purity documentation gives you confidence in your starting point.
Storage and preparation discipline protects that starting point over time.
You genuinely need both. Skipping either one undermines the other.
Storage Habits That Protect NAD+ Stability
Most peptide degradation isn’t catastrophic—it’s gradual. A vial sits out during a busy afternoon. It gets opened repeatedly. It cycles in and out of cold storage. Nothing looks wrong, but outcomes drift in ways you can’t easily diagnose.
For NAD+ peptide, the best protection is a simple, team-wide storage protocol.
Minimize exposure time
When you open the vial, treat it like focused work. Prepare what you need, seal it immediately, and return it to controlled storage. Avoid the “I’ll just leave it here while I check something” trap—those minutes add up over a study.
Avoid temperature cycling
Repeated warm-cold cycles are one of the most common and least recognized sources of peptide degradation. If your workflow involves frequent access, plan ahead. Consider preparing single-use aliquots where appropriate for your SOP, so the main stock stays undisturbed.
Standardize across your entire team
This is the step most labs underestimate. Two careful researchers with different habits create inconsistent conditions for the same compound. A shared storage protocol—documented, trained, and followed—protects your outcomes more than you might expect.
Reconstitution: The Art of Boring, Repeatable Math
The most common errors in peptide workflows are concentration errors. Not because the math is difficult, but because different people approach the same calculation differently—or don’t document it clearly enough for others to follow.
With NAD+ peptide, the goal is straightforward: choose one standard for your project and make it the team standard.
A clean approach looks like this:
Start with the labeled amount on the vial.
Choose a reconstitution volume that fits your study design.
Calculate concentration as amount divided by volume.
Document the volume and final concentration together, every single time.
The “perfect” volume matters far less than the same volume used consistently. Precision through standardization—that’s the principle.
If your team wants a shared reference for conversions and dilutions, the Peptide Calculator standardizes the math so everyone arrives at the same result using the same method.
Your Research-Ready NAD+ Workflow
If your lab is serious about reproducibility, treat intake and prep as extensions of the experiment itself—not administrative overhead.

Step 1: Receive and log
When your shipment arrives, log product name, arrival date, and lot number immediately. Store the COA in a shared, accessible location tied to that lot—no hunting, no guessing.
Step 2: Verify before first use
Match the COA lot number to the vial label. Confirm the analytical method is stated and documented. Make sure the COA meets your internal quality standards before the vial enters your workflow.
Step 3: Store immediately and consistently
Move the vial into controlled storage quickly. Avoid bench time. Don’t let “I’ll put it away in a minute” become standard practice—it will, and it matters.
Step 4: Prepare using one lab standard
Select a standard reconstitution volume for your project’s NAD+ peptide work and commit to it. If another project needs a different concentration, treat it as a separate preparation batch with clear, explicit labeling.
Step 5: Track usage across experiments
Record which lot and which preparation batch were used for every run. When outcomes drift, this record lets you immediately check whether the change aligns with a new lot, a new prep date, or altered storage access patterns.
This workflow isn’t complex. It’s consistent—and consistency is what protects your data integrity.
NAD+ in Longevity-Focused Research Programs
Many labs working with NAD+ peptide also explore adjacent compounds under the broader cellular maintenance umbrella. If that describes your program, the key is keeping workflows separated and clearly documented so assumptions don’t leak between products.
For example, if your research includes longevity-oriented peptides, maintain consistent documentation and handling standards across products like Epitalon 50mg and GHK-Cu 100mg. Different compounds, same reliability rules: log the lot, verify the COA, store consistently, prepare consistently, track what was used.
To keep sourcing standardized across your program, browse the full Peptides catalog and maintain consistent product naming in your internal records.
Troubleshooting: Quick Checks Before You Redesign
Before you assume your model or protocol is failing, run through these basics:
Was the reconstitution volume identical across all runs?
Was NAD+ peptide prepared by different people using undocumented assumptions?
Did the vial experience repeated warm-cold cycling?
Did a lot change occur without being recorded?
Were concentrations documented in consistent units across the team?
Most labs find their answer here—not in the assay. Fixing intake discipline is almost always faster than redesigning your experiment.
Wrapping Up: Treat NAD+ as a Controlled Input
The labs that get the cleanest outcomes with NAD+ peptide aren’t doing anything exotic. They’re executing fundamentals with discipline: verifying the COA, logging the lot, storing with intention, and using shared preparation standards so concentrations don’t drift between researchers.
Start with NAD+ 500MG, standardize your calculations with the Peptide Calculator, and keep your logging tight. When your input stays stable, your results become dramatically easier to interpret and reproduce.
Research Use Disclaimer: NAD+ peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How should I store NAD+ peptide for maximum stability?
Keep NAD+ peptide in controlled cold storage with minimal bench exposure. Avoid repeated temperature cycling by planning your access, and always return the vial to storage immediately after use. Standardize these habits across your entire team for best results.
What should I check on a NAD+ COA before using the peptide?
Verify three things: the lot number matches your vial label, the analytical method is clearly stated, and the documentation is lot-specific rather than generic. These checks take under five minutes and can save weeks of troubleshooting later.
How do I keep NAD+ reconstitution consistent across multiple researchers?
Choose one standard reconstitution volume for your project and document it in your SOP. Require that every preparation log includes both volume and final concentration together. Using a shared Peptide Calculator reference ensures everyone performs conversions the same way.
Epitalon Research: Quality Checks & Handling Guide
Longevity-focused research is full of subtle signals. That is both its beauty and its challenge. When your readouts are nuanced, your inputs must be rock-solid. If a vial is handled differently in week three than it was in week one, you can spend months chasing “effects” that were really just workflow drift.
That is why Epitalon research should start with the fundamentals: lot traceability, documentation you can defend, consistent storage, and preparation standards that do not change depending on who is at the bench. Once those pieces are locked in, this telomerase peptide becomes a predictable input, and your model becomes significantly easier to interpret.
If you are sourcing this compound, begin with Epitalon 50mg and build your intake routine around traceability from day one.
What Epitalon Means in a Research Workflow
In research discussions, Epithalon peptide is frequently mentioned in aging and cellular maintenance models where teams evaluate shifts in markers over time. Your specific study goals will vary, but the workflow requirement never changes: your input must be consistent across every run.
With Epitalon peptide, consistency comes from your process, not from assumptions. Your team should be able to answer these questions quickly and confidently:
- Which lot did we use?
- Where is the COA for that lot?
- What concentration did we prepare, and what volume did we use?
- How was the vial stored and accessed between runs?
If you can answer those without guessing, you can troubleshoot fast and trust your comparisons. For labs managing multiple compounds, keep a centralized product reference using Peptides for consistent naming and inventory.
Why Labs See Drift with Epitalon in Real Life
Most drift does not look dramatic. It looks like “the trend is weaker this month” or “why did this replicate shift?” and suddenly everyone is debating biology. Often, the real cause is much simpler:
- A different reconstitution volume was used by a different researcher.
- A stock was labeled loosely, so someone assumed the concentration.
- The vial was accessed more frequently during a busy week, increasing temperature cycling.
- A new lot was introduced but never recorded in the experiment notes.
That is why Epitalon pineal gland research benefits from one shared SOP that everyone follows. A good SOP does not slow your lab down. It removes the preventable variables that waste time later.
COA Review: The Intake Habit That Protects Your Study
A Certificate of Analysis is part of your experiment record, not just a compliance file. Before you prepare Epitalon peptide, confirm the COA matches the vial and contains the key details your team needs for traceability.
Lot Number Match Is Non-Negotiable
Confirm the lot or batch number on the vial matches the COA exactly. If it does not match, stop and resolve it before the vial enters your workflow. Without lot traceability, long-term comparisons become guesswork.
Analytical Method Should Be Clearly Stated
Purity only means something when it is tied to a stated method. Many peptide COAs reference HPLC profiling. Your goal is not to overanalyze the chemistry. Your goal is to confirm the method is stated clearly enough to log consistently for your records.
Lot-Specific Documentation Matters
A COA should look lot-specific, not generic. Clear documentation is what lets you quickly confirm whether a change in results aligns with a lot change.
Keep the same COA discipline across your entire inventory, whether you are logging Epitalon peptide, NAD+ 500mg, or GHK-Cu 100mg.
Purity in Practical Terms: What Quality Really Means
Purity is not just a number on a page. In real workflows, purity and stability directly influence repeatability. Impurities and degradation products can introduce background noise, and in subtle longevity models, that noise can look like a real effect.
With Epitalon peptide, quality comes down to two essentials:
- Verification of what arrived
- Protection of what arrived through consistent handling
Even high-quality material can become inconsistent if it is repeatedly warmed and cooled, left exposed during prep, or prepared at different concentrations across researchers. Think of purity verification as your baseline confidence and your SOP as the system that protects that baseline over time.
Storage and Handling: Small Habits That Protect Stability
Most peptide stability issues are caused by predictable habits: too much bench time, too much exposure, and too much temperature cycling. The fix is straightforward.
Keep Bench Time Short
Open the vial only when needed, work efficiently, seal it, and return it to controlled storage quickly. Avoid leaving it out while you handle unrelated tasks. This is one of the easiest ways to protect Epitalon peptide consistency over time.
Reduce Repeated Warm-Cool Cycles
Repeated temperature swings increase gradual degradation risk. If repeated use is expected, structure your workflow so the same container is not constantly warmed, opened, and returned. Many labs reduce cycling by preparing a controlled stock under one documented routine, then working from smaller portions. The method is less important than the consistency.
Standardize Storage Habits Across the Team
Two careful researchers can still create drift if their habits differ. Shared inventory needs shared access behavior. When storage and access behavior is standardized, Epitalon peptide becomes easier to use across long timelines without creeping variability.
Preparation and Concentration: Where Most Labs Drift
The most common “peptide problem” is concentration drift. Not because the math is hard, but because documentation is incomplete.
One person reconstitutes using one volume. Another uses a different volume. Someone logs in different units. Now two runs meant to match do not match.
For Epitalon research, choose one standard reconstitution volume for the project and stick to it. Then record volume and concentration together in the same format every time.
A clean prep record includes:
- Reconstitution volume
- Final concentration
- Prep date
- Lot number
- Initials of preparer
If your team wants a shared conversion standard, use Peptide Calculator as your common reference so everyone calculates the same way and logs results consistently.
A Repeatable Workflow Your Team Can Follow
This workflow keeps research clean without adding unnecessary friction.
Step 1: Receive and Log
Log arrival date, product name, and lot number on the day it arrives. Store the COA with the lot record so any team member can retrieve it instantly. Use the product page as your naming reference: Epitalon 50mg.
Step 2: Verify Before First Use
Match the COA lot number to the vial label. Confirm the analytical method is stated and the document looks lot-specific.
Step 3: Store Immediately and Consistently
Move the vial into controlled storage quickly. Keep bench time short during prep. Keep access behavior consistent across team members.
Step 4: Prepare Using One Lab Standard
Pick a standard reconstitution volume for Epitalon peptide for the project and do not improvise mid-study. If another project needs a different concentration, treat it as a separate preparation batch and label it clearly.
Step 5: Track Usage Across Runs
Record lot number and preparation batch details in each run’s notes. If results drift, you can quickly check whether the shift aligns with a lot change, a prep change, or a storage access pattern. That turns troubleshooting from a debate into a quick check.

Where Epitalon Fits Within a Longevity-Focused Program
Most labs do not run one compound in isolation. They build an inventory aligned to specific research themes. In longevity and cellular maintenance programs, Epitalon peptide is often stocked alongside other compounds used for broader pathway exploration.
Some labs keep energy and cellular support inputs like NAD+ 500mg in a separate workflow track, and tissue-response products like GHK-Cu 100mg under their own preparation standards.
The key point is not whether these products are “related” in conversation. The key is that each product is treated as a separate controlled input, with its own lot tracking and prep record. For a centralized inventory reference, use Peptides.
Common Mistakes That Quietly Ruin Comparability
If Epitalon peptide results start to look inconsistent, check these before rewriting your protocol:
- Did the reconstitution volume change between runs?
- Did the lot number change without being recorded?
- Was the vial accessed more often than usual, increasing temperature cycling?
- Were concentrations logged in inconsistent units or formats?
- Did different researchers handle the vial with different bench-time habits?
Most labs find the issue right here. Fixing intake and prep discipline is usually faster than redesigning the science.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Choose one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference helps keep conversions consistent.
Why does lot tracking matter so much for long projects?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly check whether the shift aligns with a lot change, saving weeks of troubleshooting.
Where should new team members look to understand what we stock?
Use Peptides as the centralized inventory list so naming and sourcing stay consistent across the lab.
Research Use Disclaimer: Epitalon (Epithalon) peptide is sold strictly for research and laboratory use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should consult all applicable institutional guidelines and regulations before use.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Choose one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference helps keep conversions consistent.
Why does lot tracking matter so much for long projects?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly check whether the shift aligns with a lot change, saving weeks of troubleshooting.
Where should new team members look to understand what we stock?
Use the Peptides catalog as the centralized inventory list so naming and sourcing stay consistent across the lab.
PT-141 Research: Quality Checks & Handling Guide
Most peptide projects do not fail because of the protocol. They drift because of small inconsistencies no one notices until the results stop lining up. A vial handled differently by a new team member. A stock prepared at a slightly different concentration. A lot change that never made it into the logbook. Suddenly the lab is debating biology when the real issue was the input all along.
That is why PT-141 research demands a clean routine from day one. When you log the lot, verify your documentation, store consistently, and keep preparation standards identical across every run, you remove the avoidable variables that create noise in melanocortin-pathway studies.
If you are sourcing Bremelanotide peptide for your lab, start with PT-141 Peptide (Bremelanotide) 10mg and treat it as a controlled research input from the moment it arrives.
What PT-141 Research Means in Your Workflow
In research settings, PT-141 is commonly referenced in melanocortin-pathway studies and receptor signaling models, where teams observe controlled response patterns under specific experimental conditions. While exact study designs vary, the workflow requirements remain constant: stable input, traceable lot history, and repeatable preparation.
With PT-141 peptide, your team should be able to answer these critical questions without hesitation:
- Which lot did we use for this run?
- Where is the COA for that specific lot?
- What concentration did we prepare, and when?
- How was the vial stored and accessed between runs?
When those answers are clear, troubleshooting becomes fast and comparisons actually mean something. For a centralized inventory reference and consistent product naming, use Peptides.
Why Labs See Inconsistent Results with PT-141
Inconsistency rarely announces itself. It creeps in through what we call routine drift. Here is how it typically happens:
- A different reconstitution volume gets used by a different team member.
- A label is vague, so someone assumes the concentration.
- The vial gets accessed more frequently during a busy week, increasing temperature cycling.
- A new lot arrives and enters the workflow without being recorded in the experiment notes.
Now the lab expects run A and run B to match, but they cannot, because the input was never actually the same. If you tighten intake and prep discipline for PT-141 peptide, these problems drop dramatically.
COA Review: The Intake Habit That Protects Your Study
A Certificate of Analysis is not paperwork, it is part of your research record. Before you prepare any Bremelanotide peptide, confirm the COA matches the vial and provides the traceability you will need later.
Lot Number Match
Confirm the lot or batch number on the vial matches the COA exactly. If it does not match, pause and resolve it immediately. Lot traceability is the foundation for meaningful comparisons across time.
Analytical Method Is Stated
Purity should be tied to a stated method. Most PT-141 peptide COAs reference HPLC profiling. Your goal is not to overanalyze the chemistry, it is to confirm the method is clearly documented so your team can record it consistently.
Lot-Specific Documentation
A COA should look lot-specific, not generic. Clear documentation makes troubleshooting far easier if you detect drift weeks into a study.
Keep this same COA discipline across your entire inventory, whether you are logging PT-141 peptide, Melanotan II, or Epitalon.
Purity in Practical Terms: What Quality Really Means
Purity matters because impurities and degradation products can introduce background noise that looks like inconsistent receptor response. That noise can be subtle, which makes it dangerous. Teams may interpret it as real biology when it is actually a material issue.
With PT-141 research, quality comes down to two things:
- Verification of what arrived (documentation)
- Protection of what arrived through consistent storage and preparation
Even clean material can become inconsistent if it is repeatedly warmed and cooled or handled differently from run to run.
Storage and Handling: Small Habits That Protect Stability
Most peptide instability issues stem from three culprits: excessive bench time, exposure, and temperature cycling. Here is how to prevent them.
Keep Bench Time Short
Open the vial only when needed, work efficiently, seal it, and return it to controlled storage quickly. Avoid leaving it out while you handle unrelated tasks.
Reduce Repeated Temperature Cycling
Repeated warm-cool cycles increase gradual degradation risk over time. If your team needs frequent access, consider preparing a controlled stock under one documented routine, then using smaller working portions. The exact approach should match your internal SOP. Consistency is what matters.
Standardize Storage Behavior Across the Team
Shared inventory requires shared habits. If one researcher handles the vial quickly and another leaves it out longer, the compound experiences different conditions. Standardized access behavior keeps PT-141 peptide stable across long timelines.
Preparation and Concentration: Keep It Consistent
Most labs encounter concentration drift not because the math is difficult, but because documentation is incomplete.
Choose one standard reconstitution volume for your PT-141 research project and stick to it. Then log volume and concentration together in the same format every single time.
A clean prep record includes:
- Reconstitution volume
- Final concentration
- Prep date
- Lot number
- Initials of preparer
If your team wants one shared conversion standard, use Peptide Calculator so everyone calculates the same way and logs results consistently.

A Repeatable Workflow Your Team Can Follow
Step 1: Receive and Log
Log arrival date, product name, and lot number on the day it arrives. Store the COA with the lot record. Use the product page as your naming reference: PT-141 Peptide (Bremelanotide) 10mg.
Step 2: Verify Before First Use
Match the COA lot number to the vial label. Confirm the analytical method is stated and the document looks lot-specific.
Step 3: Store Immediately and Consistently
Move the vial into controlled storage quickly. Keep bench time short. Maintain consistent access habits across the entire team.
Step 4: Prepare Using One Lab Standard
Pick a standard reconstitution volume for your melanocortin peptide project and do not improvise mid-study. If another project needs a different concentration, treat it as a separate prep batch and label it clearly.
Step 5: Track Usage Across Runs
Record lot number and prep batch details in each run’s notes. If results drift, you can quickly check whether the shift aligns with a lot change, a prep change, or a change in storage access patterns.
Avoiding Mix-Ups with Adjacent Products
PT-141 and Melanotan II often appear in similar research conversations, but they are different compounds and should never share assumptions in documentation or labeling. If your lab stocks both melanocortin peptides, keep records clearly separated and label preparations precisely using the product names.
Use Peptides as your centralized inventory list so your team pulls consistent names and links every time.
Common Mistakes That Quietly Ruin Comparability
If your PT-141 peptide outcomes start looking inconsistent, check these first:
- Did the reconstitution volume change between runs?
- Did the lot number change without being recorded?
- Was the vial accessed more often than usual, increasing temperature cycling?
- Were concentrations logged in inconsistent units or formats?
- Did different researchers handle the vial with different bench-time habits?
Fixing intake and prep discipline is almost always faster than rewriting the protocol.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Use one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference keeps conversions consistent across your entire lab.
Why does lot tracking matter so much in PT-141 research?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly determine whether the change aligns with a lot change rather than questioning your underlying biology.
What is the best way to organize our peptide inventory?
Use Peptides as your centralized inventory list so naming and sourcing stay consistent across your team, especially as your program grows.
Research Use Disclaimer: PT-141 (Bremelanotide) peptide is sold strictly for research and laboratory use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should consult all applicable institutional guidelines and regulations before use.
Frequently Asked Questions
How do we prevent concentration mistakes across team members?
Use one standard reconstitution volume and require that everyone logs volume and concentration together in the same format. Using Peptide Calculator as a shared reference keeps conversions consistent across your entire lab.
Why does lot tracking matter so much in PT-141 research?
Because it lets you compare runs cleanly. If outcomes shift, you can quickly determine whether the change aligns with a lot change rather than questioning your underlying biology.
What is the best way to organize our peptide inventory?
Use the Peptides catalog as your centralized inventory list so naming and sourcing stay consistent across your team, especially as your program grows.
GHK-Cu Research Guide: Quality Verification & Best Practices
Copper peptides can be remarkably consistent in research workflows—but only when your lab treats them like controlled inputs. The moment the process becomes casual, drift starts creeping in. Someone opens the vial longer than necessary, another researcher changes the preparation volume, and a third person assumes the old concentration because the label was vague. Sound familiar?
With GHK-Cu peptide (GHK-Cu copper tripeptide), these problems are entirely avoidable. The key is a tight intake routine, stable storage habits, and one preparation standard your entire team follows. When those elements are in place, the compound remains a predictable input and your work stays easier to interpret.
If you’re sourcing GHK-Cu for research, start with GHK-Cu 100mg and build your lab routine around traceability.
What GHK-Cu Represents in Research
In research discussions, GHK-Cu peptide is commonly explored in models related to tissue response, cellular signaling behavior, and extracellular matrix dynamics. Study details differ by lab, but the workflow reality is universal: the compound is only as reliable as your team’s documentation and handling.
With GHK-Cu, you want to answer these questions without guessing:
Which lot did we use?
Where is the COA for that specific lot?
What concentration did we prepare, and when?
How was the vial stored and accessed across all runs?
If your team can answer these quickly, your research stays clean. If not, variability has already started creeping in—and you just might not see it yet.
For labs running multiple products, standardizing inventory naming and sourcing through Peptides keeps everyone using the same product names and references.
Why GHK-Cu Results Drift in Real Labs
Most drift comes from small workflow differences that accumulate over time:
A vial sits out longer than intended during preparation.
It gets opened repeatedly in a humid environment.
Different team members use different preparation volumes.
A new lot is introduced but not tied into the experiment record.
Then results shift and people start debating the biology, when the real change was the input. If you treat GHK-Cu peptide as a controlled reagent with consistent logging, these issues drop away fast.
COA Review: The Intake Step That Protects Your Outcomes
Your Certificate of Analysis is part of your experimental record—not an afterthought. Before preparing GHK-Cu peptide, verify that the COA matches the vial and provides traceability your team can defend later.
Lot number match
Confirm the lot or batch number on the vial matches the COA. If it doesn’t match, stop and resolve it. Lot traceability is the base layer of repeatability—without it, everything else is built on uncertainty.
Analytical method is stated
Purity should be tied to a stated method. Many peptide COAs reference HPLC profiling. Your goal isn’t to overanalyze the methodology. It’s to confirm it’s stated clearly enough to log consistently and reference when needed.
Lot-specific documentation
A COA should look lot-specific, not generic. Vague paperwork creates vague records, and vague records create long troubleshooting sessions—usually at the worst possible time.
Keep this verification process consistent across your inventory whether you’re logging GHK-Cu peptide, BPC-157 Peptide, or TB-500 Peptide.
Purity in Practical Terms: Quality for Copper Peptides
Purity matters because impurities and degradation products can add background noise to your assays. With copper peptides, stability and handling discipline are especially important—small changes in exposure and preparation can create differences that look like real biological effects when they’re actually artifacts.
With GHK-Cu peptide, quality is the combination of verification and protection:
Verification confirms what arrived.
Consistent storage and preparation protect what arrived.
Even high-quality material becomes inconsistent if it’s repeatedly warmed and cooled or prepared differently by different researchers. Both sides matter.
Storage and Handling: Keeping GHK-Cu Stable
Most peptide issues trace back to three causes: excessive bench time, moisture exposure, and temperature cycling. The fix is simple and repeatable.
Keep bench time short
Open the vial only when needed, work efficiently, seal it, and return it to controlled storage. Avoid leaving it out while doing unrelated tasks—those gaps add up across a study.
Avoid repeated warm-cold cycling
Repeated temperature swings can increase gradual degradation risk. If repeated use is expected, plan your workflow so the vial isn’t constantly pulled out and returned.
Many labs reduce cycling by using a controlled stock preparation and then working from smaller portions when appropriate for their SOP. What matters is that the approach stays consistent across your team.
Standardize habits across the team
Two careful researchers can still create drift if their habits differ. Shared inventory needs shared access and storage behavior. When that’s standardized, GHK-Cu peptide stays more consistent across long timelines—and your data reflects that stability.
Preparation Math: Keep It Boring, Keep It Consistent
Most peptide variability originates from concentration drift. One person uses one reconstitution volume, another uses a different one, and the logs don’t make the difference obvious. Then you’re comparing experiments that were never truly comparable.
For GHK-Cu peptide, choose one standard reconstitution volume for your project and document it in a way nobody can misinterpret later.
A clean prep record includes:
Reconstitution volume
Final concentration
Prep date
Lot number
Initials of preparer
If your team wants one shared reference for conversions, use Peptide Calculator so everyone calculates the same way and logs results consistently.
A Repeatable Workflow Your Team Can Follow
Step 1: Receive and log
Log arrival date, product name, and lot number the day the vial arrives. Store the COA with that lot record.
Use the product page as a naming reference in your inventory: GHK-Cu 100mg.
Step 2: Verify before first use
Match the COA lot number to the vial and confirm the analytical method is stated.
Step 3: Store immediately and consistently
Move the vial into controlled storage quickly, keep bench time short, and keep access habits consistent across the team.
Step 4: Prepare using one lab standard
Pick one reconstitution volume for your project’s GHK-Cu peptide work and don’t improvise mid-study. If another project needs a different concentration, treat it as a separate preparation batch with clear, explicit labeling.
Step 5: Track usage across runs
Record lot number and preparation batch details in your experiment notes for each run. If results drift, you can quickly check whether the drift aligns with a lot change, a prep change, or a storage access pattern.

Avoiding Mix-Ups with Blends Containing GHK-Cu
If your lab also uses blend products, keep workflows clearly separated and labeled. For example, KLOW 80mg includes GHK-Cu as part of a standardized blend. A blend is not interchangeable with a single-compound vial—even if the same peptide appears in both.
If you compare them, comparisons only carry meaning when prep standards and logging are equally strict on both sides. Don’t let blend convenience compromise your documentation discipline.
Quick Diagnostic: Check These First
If GHK-Cu peptide outcomes start looking inconsistent, check these fundamentals before touching your protocol:
Did the reconstitution volume change?
Did the lot number change without being recorded?
Was the vial accessed more often than usual, increasing temperature cycling?
Were concentrations logged in inconsistent units across team members?
Did different researchers handle the vial with different bench-time habits?
Fixing intake and prep discipline is almost always faster than redesigning the protocol—and it usually solves the problem.
Wrapping Up: Stable Inputs, Clearer Results
GHK-Cu peptide research becomes easier to interpret when the lot is traceable, the COA is verified, storage habits are consistent, and preparation math is standardized. These aren’t dramatic measures. They’re the basics, executed with discipline.
Start with GHK-Cu 100mg, keep calculations consistent through Peptide Calculator, and keep inventory naming standardized via Peptides. When your inputs stay stable, your results become dramatically easier to trust and reproduce.
Research Use Disclaimer: GHK-Cu peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How do I prevent concentration mistakes with GHK-Cu across my team?
Use one standard reconstitution volume and require everyone to log volume and concentration together in the same format. Using Peptide Calculator as a shared reference keeps conversions consistent and prevents documentation errors.
Why is lot tracking so important for GHK-Cu research?
Lot tracking lets you compare runs cleanly over time. If outcomes shift, you can quickly determine whether the change aligns with a lot change rather than spending weeks troubleshooting your protocol.
What is the best way to store GHK-Cu copper peptide?
Keep bench time minimal, avoid repeated warm-cold cycling by planning your access, and standardize storage behavior across your entire team. These three habits protect copper peptide integrity more effectively than any single dramatic measure.
GLP-3RT Peptide Research: COA, Handling & Consistency
In the day-to-day reality of peptide research, the hardest challenge isn’t designing your protocol—it’s maintaining consistency. When you’re running a compound across multiple experiments, different team members, and weeks of work, tiny variations in handling quietly compound into significant outcome differences. That’s why GLP-3RT peptide research rewards labs that establish tight workflows from day one: clear lot tracking, verified COAs, and preparation routines that don’t change every time someone new steps up to the bench.
CoreVionRx offers GLP-3RT 30mg Research Peptide with multiple variants, which makes your documentation discipline even more important. When products have options, you need to be crystal clear about what was purchased, what was received, and what was used in each experimental run.
Set yourself up for success by sourcing directly from the GLP-3RT 30mg Research Peptide product page, then treat every incoming vial as a controlled research input—not just another item in the freezer.
What GLP-3RT Means for Your Research
GLP-3RT belongs to the broader category of metabolic signaling research, where labs investigate how peptide-driven pathways behave in controlled, non-clinical models. Specific study goals vary by team, but the workflow challenge is universal: you need stable, repeatable inputs so your experiment measures the biology—not the variability of your reagent.
That’s where GLP-3RT peptide demands a disciplined approach. When a run looks different from what you expected, you want to quickly rule out the mundane causes: a different lot, a different reconstitution volume, altered storage patterns, or a preparation date that nobody recorded.
For labs managing multiple compounds, the Peptides catalog provides a centralized inventory reference that keeps naming and sourcing consistent across your entire team.
Why Multi-Variant Products Demand Better Records
CoreVionRx lists GLP-3RT with “select options,” signaling multiple variants available. Even if your lab only purchases one version, your notes should always specify the exact product and variant received. Future comparisons depend on it—especially when procurement repeats and a different option gets selected unintentionally.
Build the habit of logging three things the moment your vial arrives: the product name exactly as sold, the variant or strength exactly as listed, and the lot number from the vial label. Store the COA with that lot record so nobody has to reconstruct the paper trail later.
This discipline is foundational to keeping GLP-3RT peptide work comparable across time and across researchers.
COA Review: Five Minutes Now, Weeks Saved Later
Your Certificate of Analysis isn’t paperwork to file and forget—it’s the document that makes your input defensible. Before preparing GLP-3RT peptide, confirm the COA matches your vial and contains the details you’ll need to interpret results six months from now.
Start with the lot number. The COA lot must match the vial label exactly. No exceptions. Without lot traceability, you cannot confidently compare one run to another, and that uncertainty undermines your entire dataset.
Look for the analytical method. Most peptide COAs reference HPLC-based purity profiling. You’re not trying to evaluate the lab’s methodology—you’re confirming that a method is stated, that documentation is lot-specific, and that your records will be clear enough to troubleshoot with when results shift.
If your lab already maintains COA intake standards for other CoreVionRx products, apply that same discipline to GLP-3RT peptide. Consistency in intake is one of the fastest ways to reduce long-term experimental variability.
Purity in Real-World Terms
Purity isn’t a marketing bullet point—it’s a reproducibility factor. When impurities or degradation products creep in, they add background noise to sensitive readouts. That noise doesn’t always announce itself. Sometimes it looks like a subtle trend that your team wastes hours trying to interpret biologically.
With GLP-3RT peptide, the goal isn’t perfection. It’s confidence. You want to know that your input didn’t change between the run that looked strong and the run that looked off.
That confidence comes from pairing verification with handling discipline. Verification tells you what arrived. Handling discipline protects what arrived from the moment it enters your lab.
Storage Habits That Protect Integrity
Most stability issues develop slowly. A vial left out during a busy afternoon. Pulled from cold storage repeatedly. Opened casually, then returned. Nothing looks dramatic, but outcomes drift—and you may not notice until you’re comparing datasets that should align.
For GLP-3RT peptide, the best storage habits are straightforward and realistic.
Keep bench time short. When the vial is open, treat it as focused work time. Prepare what you need, seal it, and return it to controlled storage immediately. Don’t leave it sitting while you answer emails or set up the next assay.
Minimize warm-cold cycling. If multiple uses are expected, plan your workflow so you’re not constantly removing the same container from storage. Many labs reduce cycling by preparing controlled stocks and using aliquots where appropriate for their SOP. The specific technique matters less than doing it consistently.
Standardize access behavior across your team. One researcher handles the vial quickly; another leaves it out longer. Neither is “wrong,” but the compound experiences different conditions—and that inconsistency shows up in your data. Shared habits protect shared inventory.
Preparation Math: Where Most Labs Quietly Drift
When you audit why results vary between runs, you’ll find a common culprit: concentration drift. One person reconstitutes with one volume. Another uses a different volume. Someone records the concentration in milligrams per milliliter, someone else assumes micrograms per microliter. Now two “identical” experiments aren’t identical at all.
With GLP-3RT peptide, the fix is one standard that everyone follows. Choose a reconstitution volume for your project, use it every time for that project, and document it the same way in every log entry.
A clean documentation line includes the reconstitution volume and the final concentration together. That single line prevents most misunderstandings later.
For shared conversion consistency, the Peptide Calculator page serves as a practical internal reference so your entire team calculates using the same method and the same steps.
The goal isn’t “using a calculator.” It’s ensuring GLP-3RT peptide prep stays consistent across people and across time.
A Repeatable Workflow Your Team Can Actually Follow
A good workflow doesn’t add complexity—it removes preventable variability.
Log on arrival. Record exactly what you purchased and what you received, especially important when the product has variants.
Verify the COA. Match it to the vial. Store it with the lot record so it’s always accessible.
Store immediately and consistently. Reduce bench time, reduce temperature cycling, and ensure multiple researchers follow the same storage behavior.
Prepare using one standard. One reconstitution volume for the project. If another project needs a different concentration, treat it as a separate preparation batch with clear, explicit labeling.
When you execute these steps reliably, GLP-3RT peptide becomes the stable input your research deserves.
GLP-3RT Alongside Other Metabolic Research Products
Most labs don’t work with one compound at a time. They build focused inventories aligned to study goals. In metabolic signaling programs, it’s common to keep adjacent options available for comparisons, method development, or controlled baselines.
If your lab compares against other products, maintain clean documentation and separated workflows. For example, Glp-lr3 30mg Research Peptide appears in the same CoreVionRx catalog as GLP-3RT, and it should be treated as a completely separate input with its own preparation and storage records.
Comparison only carries meaning when handling standards are identical across conditions. Different preparation volumes between compounds create hidden variables that make interpretation harder, not easier.
Quick Diagnostic Checklist
Before you assume your protocol needs redesigning, check these fundamentals:
Was the reconstitution volume identical across all GLP-3RT peptide runs?
Did the lot number change without being recorded?
Did storage access patterns shift, with more frequent warm-cold cycling?
Were concentrations recorded in inconsistent units?
Did a new researcher follow different preparation habits without realizing it?
Most labs find their answer in these questions. Fixing intake discipline is almost always faster than redesigning experiments.
Wrapping Up: Consistency Is the Secret
The labs getting the cleanest outcomes aren’t doing anything magical. They’re executing basics with consistency. GLP-3RT peptide research becomes dramatically easier to interpret when your input is traceable, your COA is verified, storage is consistent, and preparation math is standardized.
Source from GLP-3RT 30mg Research Peptide, standardize calculations using the Peptide Calculator, and keep your inventory organized through Peptides. When inputs stay stable, your results become clearer and your troubleshooting faster.
Research Use Disclaimer: GLP-3RT peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How do I keep GLP-3RT prep consistent across multiple researchers?
Choose one standard reconstitution volume for your project, require everyone to log volume and concentration in the same format, and keep calculations consistent. Using Peptide Calculator as a shared reference prevents conversion mistakes and ensures everyone follows the same method.
Why does lot tracking matter so much for GLP-3RT research?
Lot tracking lets you compare runs cleanly. If results shift, you can quickly determine whether the change aligns with a lot change—one of the most common hidden causes of variability in peptide research.
How should I handle GLP-3RT storage to maintain stability?
Keep bench time minimal, avoid repeated warm-cold cycling by planning your access, and standardize storage behavior across your entire team. These three habits protect peptide integrity more effectively than any single dramatic measure.
Thymosin Alpha-1 Research: Quality Checks & Handling Guide
Peptide research unravels when workflows become casual. A vial gets opened for “just a minute,” prep math gets done slightly differently by different people, and before you know it, your lab is comparing runs that were never truly comparable. When that happens, you can’t tell whether your model is shifting or your reagent is drifting—and that uncertainty wastes time and resources.
That’s why Thymosin Alpha-1 peptide (Thymosin α1) research should begin with a clean, repeatable routine. When your intake documentation is tight, storage habits are consistent, and preparation math is standardized, you eliminate the quiet variables that ruin experimental clarity. The point isn’t to overcomplicate your workflow. It’s to remove the obvious sources of drift before they cost you weeks of troubleshooting.
If you’re sourcing Thymosin Alpha-1 for research, start with Thymosin Alpha-1 Peptide and treat it as a controlled material from day one.
What Thymosin Alpha-1 Means in Your Research
In research contexts, Thymosin Alpha-1 is commonly explored in immune-signaling and immune-response models where teams evaluate pathway behavior under controlled conditions. The practical reality for your lab is simpler than the biology: it’s a research input that needs to stay stable and traceable across your entire study timeline.
With Thymosin Alpha-1 peptide, stability and traceability depend less on what people believe about the compound and more on how your team treats the vial:
Is the lot recorded and accessible?
Is the COA stored with that lot record?
Is the vial stored consistently and accessed with discipline?
Is the concentration prepared the same way every single time?
If you can answer these questions quickly, your research outcomes stay consistent. If you can’t, variability creeps in—and you might not notice until you’re trying to figure out why week three’s data doesn’t match week one’s.
For a centralized inventory view that keeps naming consistent, the Peptides page helps your team standardize sourcing references across all products.
Why Thymosin Alpha-1 Results Drift in Real Labs
Most inconsistency comes from ordinary workflow drift, not from dramatic “bad vial” scenarios. It’s the accumulation of small lapses:
A researcher reconstitutes using a different volume without recording it clearly.
Another researcher assumes the old concentration was the same.
The vial gets temperature-cycled more often during a busy week.
A new lot arrives and gets used before it’s tied into the study record.
Then results shift slightly and the lab debates the biology, when the real change was the input all along.
If repeatability is your goal—and it should be—Thymosin Alpha-1 peptide needs one shared SOP that everyone follows without exception.
COA Review: Five Minutes That Protect Your Project
Your Certificate of Analysis isn’t just a document to file away. It’s the paper trail that makes your reagent defensible and your troubleshooting fast.
Before you prepare Thymosin Alpha-1 peptide, verify three things.
Lot number match
Confirm the lot or batch number on the vial matches the COA. If it doesn’t match, pause and resolve it first. Without a lot match, you cannot compare results over time with confidence—and confidence in your input is everything.
Stated analytical method
Purity is only meaningful when tied to a stated method. Many peptide COAs reference HPLC-based profiling. The key isn’t to overanalyze the method. The key is that the method is stated clearly enough for your lab to record and interpret consistently.
Lot-specific documentation
A COA should look lot-specific, not generic. If documentation is vague, records become vague, and that’s where confusion grows—usually right when you can least afford it.
This COA discipline matters even more when Thymosin Alpha-1 peptide is used across multiple experiments, because it lets you quickly check whether any drift aligns with a lot change, a prep change, or a storage access pattern.
Purity in Practical Terms: What Quality Really Means
In day-to-day research, purity isn’t a marketing number—it’s a reproducibility factor. Impurities and degradation products can introduce background noise that shows up as inconsistent readouts, especially in sensitive immune-response models.
With Thymosin Alpha-1 peptide, quality is the combination of two things: verification of what arrived, and protection of what arrived through consistent handling. Even a clean lot can become inconsistent if the vial is repeatedly warmed and cooled, left exposed during prep, or handled differently by different team members.
Think of purity verification as your baseline confidence. Think of your SOP as what protects that baseline over the entire study timeline.
Storage and Handling: Keeping Thymosin Alpha-1 Stable
Most peptide issues develop slowly. The vial sits out longer than intended. It gets accessed more often than expected. It gets opened while someone is multitasking, and moisture exposure increases. Nothing looks dramatic, but outcomes drift—and that drift undermines your data.
With Thymosin Alpha-1 peptide, a few simple habits make a genuine difference.
Keep bench time short
When the vial is opened, treat it as focused work. Prepare what you need, seal it, and return it to controlled storage quickly. Avoid leaving it out while you handle unrelated tasks. Short bench time is one of the easiest ways to protect consistency.
Reduce repeated temperature cycling
Repeated warm and cold cycles can increase gradual degradation risk over time. If repeated use is expected, plan your workflow to reduce how often the same container is pulled out, warmed, opened, and returned.
Many labs handle this by preparing a controlled stock under one documented routine and then using an access approach that minimizes repeated cycling of the same container. The exact method should match your internal SOP. What matters is that it stays consistent.
Standardize storage behavior across the team
Two careful researchers can still create drift if their habits are different. Shared inventory needs shared habits. When access behavior is standardized, Thymosin Alpha-1 peptide becomes easier to run across long timelines without creeping variability.
Preparation Math: Where Labs Drift Without Realizing
The most common “peptide problem” in real labs is concentration drift. One person reconstitutes using one volume. Another person uses a different volume. Someone logs the concentration in a different unit. Now two runs meant to match don’t match—and the discrepancy is buried in poor documentation.
For Thymosin Alpha-1 peptide, the fix is simple: choose one standard reconstitution volume for your project and stick to it. No exceptions.
A clean preparation record includes:
Reconstitution volume
Final concentration
Prep date
Lot number
Initials of the preparer
That single line removes most assumptions—and most sources of error.
If your team wants a shared reference for dilution and conversion math, use Peptide Calculator so everyone calculates the same way using the same method.
The goal isn’t the calculator itself. The goal is consistent math and consistent logging across your entire team.
A Repeatable Workflow Your Team Can Actually Follow
This workflow keeps your research clean without adding unnecessary complexity.
Step 1: Receive and log
Log arrival date, product name, and lot number on the day the vial arrives. Store the COA with that lot record so any team member can find it instantly.
Use the product page link in your inventory record so naming stays consistent: Thymosin Alpha-1 Peptide.
Step 2: Verify before first use
Match the COA lot number to the vial label and confirm the analytical method is stated. Confirm the COA looks lot-specific rather than generic.
Step 3: Store immediately and consistently
Move the vial into controlled storage quickly. Keep bench time short during prep. Keep access behavior consistent across team members.
Step 4: Prepare using one lab standard
Pick a standard reconstitution volume for Thymosin Alpha-1 peptide for your project and don’t improvise mid-study. If another project needs a different concentration, treat it as a separate preparation batch with clear labeling so nobody assumes the wrong standard later.
Step 5: Track usage across runs
Record lot number and prep batch details in each run’s experiment notes. If results drift, you can immediately check whether the drift aligns with a lot change, a prep change, or a change in storage access patterns.
This keeps troubleshooting quick and prevents wasted cycles.
Thymosin Alpha-1 Alongside Related Products
Many labs keep a focused peptide inventory aligned to specific research themes. The important part isn’t whether products are “related” in conversation—it’s that each product is treated as a separate controlled input with its own lot tracking and preparation record.
If your lab also uses inflammation and recovery-adjacent products like TB-500 Peptide (Thymosin Beta-4) or BPC-157 Peptide, keep workflows clearly separated and labeled. Don’t let assumptions about one compound’s preparation bleed into another compound’s SOP.
If you prefer a centralized inventory view for ordering and naming consistency, keep your internal reference tied to Peptides so the whole team uses the same product naming and links.
Quick Diagnostic: Before You Touch the Protocol
If your Thymosin Alpha-1 peptide results start looking inconsistent, check these before redesigning anything:
Did the reconstitution volume change between runs?
Did the lot number change without being recorded?
Was the vial accessed more often than usual, increasing temperature cycling?
Were concentrations documented in inconsistent units across team members?
Did different researchers handle the vial with different bench-time habits?
Most labs find the issue right here. Fixing intake and prep discipline is usually faster—and always cheaper—than redesigning the science.
Wrapping Up: Stable Inputs, Clear Results
The labs that get the cleanest outcomes aren’t doing anything complicated. They’re doing the basics consistently. Thymosin Alpha-1 peptide research becomes easier to interpret when the lot is traceable, the COA is verified, storage habits are consistent, and preparation math is standardized across the team.
Start with Thymosin Alpha-1 Peptide, keep conversions consistent through Peptide Calculator, and keep inventory naming standardized via Peptides. When inputs stay stable, your results become clearer and your troubleshooting becomes dramatically faster.
Research Use Disclaimer: Thymosin Alpha-1 peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How do I keep Thymosin Alpha-1 prep consistent across multiple researchers?
Choose one standard reconstitution volume for your project and require everyone to log volume and concentration in the same format. Using Peptide Calculator as a shared reference helps prevent conversion drift between team members.
Why does lot tracking matter so much for Thymosin Alpha-1 research?
Lot tracking lets you compare runs cleanly over time. If results shift, you can quickly check whether the shift aligns with a lot change—one of the most common hidden causes of variability in immune peptide research.
What storage conditions work best for Thymosin Alpha-1 peptide?
Keep bench time minimal, avoid repeated warm-cold cycling by planning your access patterns, and standardize storage behavior across your entire team. These habits protect peptide integrity more effectively than any single measure.
CJC-1295 Ipamorelin Research: Quality Checks & Handling
Peptide blends are convenient, but they magnify workflow errors. With a single compound, one mistake might be obvious. With a blend, mistakes hide inside assumptions. Someone preps using a different volume, someone else labels it loosely, and now your supposedly identical setup across runs isn’t identical at all.
That’s why CJC-1295 + Ipamorelin peptide research demands discipline from day one—not eventually, not when problems appear, but from the first vial you open. You need to trace the lot, verify documentation, store consistently, and prepare the same concentration every single time. When those fundamentals are locked in, the blend becomes a stable input and your study can focus on what it’s actually designed to measure.
If you’re sourcing this growth hormone secretagogue blend, start with the CJC-1295 + Ipamorelin 10mg product page and treat it as a controlled material from the moment it arrives.
What This Blend Means in Your Research Workflow
In research contexts, CJC-1295 and Ipamorelin are commonly referenced in growth-hormone-axis signaling studies and related experimental models. The blend format lets researchers observe pairing behavior under a consistent setup. The lab reality is straightforward: a blend reduces preparation steps, but it increases the importance of meticulous recordkeeping.
With CJC-1295 + Ipamorelin peptide, your team should be able to answer these questions without hunting through notebooks:
Which lot did we use?
Where is the COA for that exact lot?
What volume did we reconstitute with?
What concentration did we label and log?
How was the vial stored and accessed between runs?
If your team can answer these quickly, troubleshooting stays simple. If not, you’re flying blind.
For inventory consistency across your peptide program, the Peptides page keeps product naming and sourcing standardized.
Why Blends Create More Variability Than Single Compounds
Most variability doesn’t come from dramatic failures—it comes from small workflow drift that accumulates when different people handle the same material.
Someone reconstitutes with a different volume and doesn’t record it clearly.
Someone uses a vague label like “CJC/IPA stock” instead of logging the exact concentration.
The vial gets pulled from controlled storage repeatedly during a busy run week.
A new lot arrives and gets used without being tied to the experiment record.
Then, when results shift, you waste time debating whether biology changed when the real change was the input all along.
That’s why CJC-1295 + Ipamorelin peptide needs tighter habits than a single-compound workflow ever would.
COA Review: Your Five-Minute Quality Gate
A Certificate of Analysis isn’t a formality—it’s part of your experimental record. Before any prep, verify that the COA matches the vial and gives you traceability you can defend six months from now.
1) Lot number match is non-negotiable
Confirm the lot or batch number on the vial matches the COA exactly. If it doesn’t match, stop and resolve it. Without lot traceability, comparing runs across time becomes guesswork, and guesswork isn’t research.
2) The analytical method should be stated
Purity only carries meaning when tied to a stated method. Many peptide COAs reference HPLC profiling for purity verification. Your goal isn’t to become an analytical chemist—it’s to confirm the method is stated and documented clearly enough that your lab can record it consistently.
3) The document should look lot-specific
A COA should feel tied to the exact lot you received, not like a generic template that could apply to any vial. Lot-specific documentation makes troubleshooting faster when results drift later—and they sometimes do.
This is especially important for CJC-1295 + Ipamorelin peptide because you’re using one vial to represent two distinct inputs. Your documentation is what prevents confusion down the road.
Purity in Practical Terms: Quality for Blends
Purity matters for the same reason it matters with any research material: it supports repeatability. Impurities or degradation products can add background noise that masquerades as inconsistent signaling or variable response. The most frustrating part? That noise can look exactly like real biology.
With CJC-1295 + Ipamorelin peptide, purity is only half the equation. The other half is what happens after the vial arrives. Even a pristine material becomes inconsistent if your lab repeatedly warms and cools it, leaves it exposed during prep, or prepares it at different concentrations depending on who’s at the bench.
Think of purity verification as your baseline confidence, and your SOP as what actively protects that baseline.
Storage and Handling: Protecting Blend Stability
The most common storage mistakes aren’t dramatic—they’re gradual.
The vial stays out too long during prep.
It gets temperature-cycled more often than anyone tracks.
Multiple team members access it with different habits and different bench-time behavior.
With CJC-1295 + Ipamorelin peptide, your goal is keeping handling consistent so the input doesn’t change between week one and week four.
Keep exposure time short
When you open the vial, treat it as focused work. Prepare what you need, seal it, and return it to controlled storage. Avoid leaving it out while handling unrelated tasks. Short bench time is one of the easiest ways to protect stability.
Avoid repeated warm-cold cycling
Repeatedly removing the same vial from cold storage, letting it warm, opening it, and returning it can increase gradual degradation risk. If repeated use is expected, plan your workflow to reduce how many times the same container is cycled.
A practical approach is preparing a controlled stock under one documented routine, then working from smaller portions when appropriate for your SOP. The specific method matters less than doing it the same way every time.
Standardize storage behavior across the team
Two careful researchers can still create drift if their habits differ. Shared inventory needs shared habits. When storage and access behavior is standardized, CJC-1295 + Ipamorelin peptide becomes easier to run across longer timelines without creeping variability.
Preparation Math: Where Blends Introduce Extra Risk
Most peptide problems are concentration problems wearing a different costume. Not because the math is difficult, but because documentation is inconsistent.
One person writes “reconstituted” without recording the volume.
Another person assumes the old volume.
A third person logs units differently.
Now two experiments meant to match don’t match—and nobody knows why.
For CJC-1295 + Ipamorelin peptide, choose a standard reconstitution volume for your project and commit to it. Then document it in a way that no one can misinterpret later.
A clean prep log line includes:
Reconstitution volume
Final concentration
Prep date
Lot number
Initials of preparer
That one line removes most assumptions—and most errors.
If your team wants a shared reference for conversions, the Peptide Calculator keeps the method consistent even when the person doing the prep changes.
A Repeatable Workflow for Your Team
This workflow keeps your setup clean without adding unnecessary friction.
Step 1: Receive and log
Log arrival date, product name, and lot number the day the vial arrives. Save the COA with the lot record so anyone can retrieve it without digging.
Step 2: Verify before first use
Match the COA lot number to the vial label. Confirm the analytical method is stated. Ensure the COA looks lot-specific.
Step 3: Store immediately and consistently
Move the vial into controlled storage quickly. Avoid long bench time. Keep access behavior consistent across your team.
Step 4: Prepare using one standard
Pick a reconstitution volume standard for your project’s CJC-1295 + Ipamorelin peptide work and don’t improvise mid-study. If another project needs a different concentration, treat it as a separate prep batch with clear labeling.
Step 5: Track usage across runs
Record which lot and prep batch was used in each run. If results drift, you can immediately check whether the drift aligns with a lot change, a prep change, or a storage access pattern. This turns troubleshooting from a debate into a quick check.
Avoiding Mix-Ups with Related Products
Many labs run multiple peptides under a shared procurement program. That’s efficient, but it increases the risk of assumption drift if labeling and logs aren’t strict.
If your lab also stocks Tirzepatide or PT-141, keep them logged as separate inputs with separate prep standards and separate batch records. The similarity is in how people talk about them, not in how you should document them.
To keep procurement organized and naming consistent across your team, maintain one shared inventory reference using Peptides so everyone pulls the same product names and links.
Quick Diagnostics: Before You Assume the Protocol Failed
If your results start looking inconsistent, check these fundamentals before redesigning anything:
Did the reconstitution volume change between runs?
Did the lot number change without being recorded?
Was the vial accessed more often than usual, increasing temperature cycling?
Did multiple researchers label stocks differently?
Were concentrations recorded in inconsistent units or formats?
Most labs find the issue right here. Fixing intake and prep discipline is almost always faster than redesigning the entire experiment.
Wrapping Up: Keep the Blend Stable, Keep Your Data Clean
Blends are only “easy” when the workflow is strict. CJC-1295 + Ipamorelin peptide becomes a stable research input when the lot is traceable, the COA is verified, storage is consistent, and preparation math is standardized across your team.
For the cleanest path, source from CJC-1295 + Ipamorelin 10mg, standardize conversions with the Peptide Calculator, and keep inventory references consistent through Peptides. When your input stays stable, your results become easier to interpret and far easier to reproduce.

Research Use Disclaimer: CJC-1295 + Ipamorelin peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How do I keep CJC-1295 + Ipamorelin prep consistent across multiple researchers?
Choose one standard reconstitution volume for your project, require everyone to log volume and concentration in the same format, and store the COA with the lot record. Using Peptide Calculator as a shared conversion reference prevents math drift between team members.
Why does lot tracking matter more for a peptide blend?
Because you are using one vial to represent two distinct inputs. If results drift, lot tracking is the fastest way to confirm whether the input changed, helping you isolate variables efficiently.
What is the best way to store CJC-1295 + Ipamorelin peptide?
Keep bench time minimal, avoid repeated warm-cold cycling by planning your access, and standardize storage behavior across your entire team. These habits protect blend stability more effectively than any single dramatic measure.
BPC-157 Research: Quality Verification & Handling Guide
When labs say a peptide “didn’t work,” what they usually mean is the workflow wasn’t consistent. The compound itself is likely fine, but somewhere between receipt and assay, the prep volume changed, the vial got temperature-cycled too often, or a new lot slipped into the rotation without being documented. Over a multi-week project, these small differences stack into noise that looks like biology.
That’s precisely why BPC-157 peptide research benefits enormously from a tight, repeatable intake and preparation routine. When your workflow is locked down, you can focus on your model and your protocol instead of constantly wondering whether the input drifted.
If you’re sourcing BPC-157, start from BPC-157 Peptide and treat it like a controlled research input from the moment it arrives at your lab.
What BPC-157 Means in Your Research Workflow
In research settings, BPC-157 is commonly discussed in tissue-response and repair-adjacent models where labs track cellular signaling changes under carefully controlled conditions. The important part for your workflow isn’t the compound’s reputation—it’s that your lab can standardize BPC-157 like any other reagent: lot tracked, documentation stored, storage conditions consistent, and concentration preparation fully repeatable.
That mindset—treating BPC-157 peptide as a stable input rather than a variable—is what separates reproducible results from frustrating inconsistency.
For labs managing multiple compounds, keeping everything organized through the broader Peptides catalog ensures your team uses consistent naming and purchasing references across every project.
Why BPC-157 Results Drift in Real Labs
Most drift isn’t dramatic. It happens because busy teams rely on memory instead of records.
A researcher reconstitutes using a different volume because it “felt right.”
A teammate assumes the older concentration without checking.
The vial gets pulled from storage more frequently during a heavy week of runs.
A new lot arrives, but the lot number never makes it into the experiment notes.
Now you’re comparing run A to run B and expecting them to match, but they can’t—the input wasn’t actually the same. When you tighten the workflow around BPC-157 peptide, these issues disappear fast.
COA Review: Five Minutes That Prevents Weeks of Confusion
Your Certificate of Analysis isn’t just a compliance file—it’s part of your research record. Before preparing BPC-157 peptide, verify the COA against the vial and log what matters.
1) Lot or batch number must match the vial
Start here. The COA lot should match the vial label exactly. If it doesn’t match, stop and resolve it before proceeding. Lot traceability is the foundation of repeatable work—everything else builds on this.
2) The COA should state the analytical method
Purity only carries meaning when it’s tied to a stated method. Many peptide COAs reference HPLC profiling. Whatever method appears, your goal is simple: confirm it exists, confirm it’s stated clearly, and log it consistently in your records so you can reference it later.
3) The COA should feel lot-specific
A COA should look like it belongs to your specific lot, not like a generic template that could apply to anything. Lot-specific documentation makes your research defensible and makes troubleshooting dramatically faster when you need it.
This COA habit matters especially for BPC-157 peptide because it’s often used across many runs spanning weeks or months. The longer your timeline, the more valuable lot tracking becomes.
Purity in Practice: A Reproducibility Factor
In day-to-day research, purity isn’t a marketing number—it’s a practical reproducibility factor. Impurities or degradation products can add background noise to assays, particularly when your readouts are sensitive or your study design relies on detecting subtle shifts over time.
With BPC-157 peptide, the goal isn’t “perfect.” The goal is “consistent enough that changes in your results actually reflect the experiment, not the input.”
Think of it this way:
Purity documentation helps you trust what arrived.
Handling discipline protects what arrived from degradation.
If either side is weak, your outcomes can drift—and you might not notice until it’s too late.
Storage and Handling: Habits That Keep Inputs Stable
Most peptide instability stems from predictable workflow habits: too much bench time, too much exposure, and too many warm-cold cycles. The good news? These are all preventable.
Keep the vial dry and exposure minimal
Lyophilized peptides support stability, but only when handled correctly. Open the vial only when needed, work efficiently, seal it, and return it to controlled storage quickly. Avoid leaving it on the bench while switching tasks—those transitions add up.
Reduce repeated temperature cycling
Repeatedly pulling a vial from controlled storage, letting it warm, opening it, and returning it creates gradual degradation risk. If repeated use is expected, structure your workflow so you’re not constantly cycling the same container.
Many labs reduce cycling by planning the week’s work in advance or using an aliquot approach that fits their internal SOP. There isn’t one “perfect” method—there’s only the method your team follows consistently.
Standardize storage behavior across your whole team
This is where labs quietly lose consistency. Two researchers with different habits expose the same compound to different conditions. For shared inventory, shared habits protect shared outcomes—and that standardization pays dividends across long studies.
Preparation and Concentration: Where Most Labs Drift
If you audit peptide problems across labs, a surprising percentage trace back to concentration issues. Not because the math is hard—because different people do the math differently, or don’t record it clearly enough for others to interpret.
For BPC-157 peptide, choose one standard for your project and make everyone follow it.
A clean standard looks like this:
Start with the labeled amount.
Pick a reconstitution volume that fits your study design.
Concentration equals amount divided by volume.
Document the volume and final concentration together, every single time.
That last step is the key. When volume and concentration are always recorded together, assumptions—and the errors they create—simply disappear.
If your team wants a shared reference for dilution math, the Peptide Calculator serves as your standard conversion tool. The tool itself isn’t the point. Consistency is.
A Repeatable BPC-157 Workflow Your Team Can Follow
A good workflow reduces friction and prevents avoidable drift without adding bureaucracy.
Step 1: Receive and log
Log arrival date, product name, and lot number the same day the vial arrives. Store the COA with the lot record so anyone can find it quickly without guessing.
Step 2: Verify documentation before first use
Match the COA lot number to the vial label. Confirm the analytical method is stated. Make sure the document is clear enough for your internal standards.
Step 3: Store immediately and consistently
Move the vial into controlled storage quickly. Avoid leaving it out during unrelated tasks. Consistency matters far more than perfection here.
Step 4: Prepare using one lab standard
Pick one reconstitution volume for your project’s BPC-157 peptide work and stick to it. If another project needs a different concentration, treat it as a separate preparation batch with clear, explicit labeling so nobody assumes the wrong standard later.
Step 5: Track which batch was used in which run
Record lot number and prep date in the experiment record for each run. If outcomes drift, you can instantly check whether the drift aligns with a new lot, a new prep date, or a change in access behavior.
This routine keeps BPC-157 peptide as a stable input rather than a hidden variable in your research.
BPC-157 Alongside Related Peptides in Your Inventory
Many labs running tissue-response studies keep a focused group of peptides in rotation. The key isn’t whether products are “related” in conversation—it’s that your lab maintains consistent documentation and handling standards across all of them.
If your inventory includes TB-500 Peptide (Thymosin Beta-4), keep it logged and prepared as a separate input with separate prep records. If your lab uses blend products containing BPC-157 peptide, keep those workflows clearly separated too so assumptions don’t leak between runs.
For example, KLOW 80 mg includes BPC-157 as part of a standardized blend, and GLOW 70 mg is another blend format. A blend is not interchangeable with a single vial. If your lab compares them, the comparison only carries weight when storage, prep standards, and documentation are equally strict on both sides.
Diagnostic Checklist: Before You Redesign
If your BPC-157 peptide results start looking inconsistent, run through these before touching your protocol:
Was the reconstitution volume identical across runs?
Did the lot number change without being recorded?
Did the vial experience more warm-cold cycling than usual?
Were concentrations recorded in consistent units across researchers?
Did multiple people handle the vial with different bench-time habits?
Most labs find their issue right here. Fixing intake and prep discipline is usually faster—and far less disruptive—than rewriting your science.

Wrapping Up: Stable Inputs, Clean Data
The best way to protect your research is to protect your inputs. BPC-157 peptide work becomes far easier to interpret when the lot is traceable, the COA is verified, storage habits are consistent, and preparation math is standardized across your team.
Start with BPC-157 Peptide, keep your inventory organized through Peptides, and standardize conversion math using the Peptide Calculator. When your workflow stays consistent, your outcomes become clearer and your troubleshooting becomes much faster.
Research Use Disclaimer: BPC-157 peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How do I verify BPC-157 purity before using it in research?
Verify three things on your COA: the lot number matches your vial label, the analytical method is clearly stated, and the documentation is lot-specific rather than generic. These checks ensure you are working with a documented, traceable input.
How can I prevent concentration mistakes across my research team?
Choose one standard reconstitution volume for your project and require everyone to log volume and concentration together in the same format. Using Peptide Calculator as a shared reference keeps conversions consistent across all team members.
Why is lot tracking important if the BPC-157 product name is the same?
Even with the same product name, different lots can vary slightly. If results drift, lot tracking is the fastest way to separate whether biology changed or input changed, saving you from unnecessary protocol redesigns.
TB-500 Peptide Research Guide: Quality Checks & Handling
Plenty of peptide projects don’t stumble because the research question is weak—they falter because the workflow became inconsistent. One researcher prepares a vial one way, another assumes a different concentration three weeks later, and suddenly you’re debating results that were never truly comparable. With TB-500 peptide (also known as Thymosin Beta-4 fragment), that kind of drift is entirely avoidable—if your team treats the compound like a controlled input from the moment it arrives.
This guide cuts straight to what matters for repeatability: confirming lot documentation, protecting integrity through storage and handling, and standardizing reconstitution math so every preparation matches the next without guesswork.
If you’re sourcing TB-500, start with TB-500 Peptide (Thymosin Beta-4) and build your intake and preparation routine around traceability.
What TB-500 Represents in Research Settings
TB-500 is commonly referenced as a synthetic peptide related to thymosin beta-4 fragments, frequently studied in models examining tissue response, cellular activity, and wound healing pathways. In practical lab terms, the value isn’t the label—it’s that TB-500 can serve as a defined, consistent research material when you control the variables that teams often forget to manage.
That’s why TB-500 peptide research thrives when your lab can answer these four questions quickly:
Which lot did we use, and where is the record?
Where is the COA for that exact lot?
What concentration did we prepare, and what reconstitution volume did we use?
How was the vial stored and accessed throughout the study timeline?
If your team can answer these without hunting through notebooks, troubleshooting stays simple. If not, you’re troubleshooting in the dark.
Why Workflow Discipline Matters More Than Most Labs Admit
Peptide variability rarely announces itself. It appears as small outcome shifts that seem “interesting” at first and frustrating later. With TB-500 peptide, the most common sources of drift aren’t mysterious—they’re everyday workflow issues:
A vial sits out longer than it should during preparation.
The same vial gets pulled from controlled storage repeatedly, warming and cooling many times.
Two researchers reconstitute using different volumes without clear documentation.
A new lot arrives and gets used without being tied to the experiment record.
The fix isn’t complex. It’s a standard intake routine and a standard prep routine that everyone follows—no improvisation, no assumptions.
COA Review: Your Pre-Flight Checklist
A Certificate of Analysis isn’t administrative paperwork. It’s part of your experimental record. Before you prepare TB-500 peptide, confirm the COA matches the vial and includes the traceability details your lab depends on.
Lot number matching is non-negotiable
Start with the lot or batch number. The COA lot must match the vial label exactly. If it doesn’t match, pause and resolve it before doing anything else. Without lot traceability, comparing runs across time becomes speculation—and speculation isn’t science.
Confirm the analytical method is stated
Purity is only meaningful when tied to a stated analytical method. Many peptide COAs reference HPLC profiling for purity verification. Your goal isn’t to critique the method—it’s to confirm a method is clearly documented so your lab can record it consistently and interpret the purity value the same way every time.
Ensure the document is lot-specific
A COA should look like it belongs to that specific lot. Generic documentation creates generic records, and generic records breed confusion when you’re trying to troubleshoot six months later.
If your lab already follows COA intake standards for other products, apply that same routine here. The process should be identical whether you’re logging TB-500 peptide, BPC-157 Peptide, or GHK-Cu 100mg.
Purity: What “Quality” Really Means in Your Lab
In real research workflows, purity isn’t a specification to brag about—it’s a reproducibility factor. Impurities or degradation products can introduce background noise that masquerades as inconsistent biology. That noise can be subtle, which makes it dangerous, because teams may spend days interpreting patterns that were actually created by input variability.
With TB-500 peptide, the goal is confidence in your starting point and protection of that starting point through disciplined handling. Even high-quality material becomes inconsistent if it’s repeatedly exposed to humidity, temperature fluctuations, or different preparation approaches.
Think of purity verification as your baseline confidence—and your SOP as the system that preserves that baseline across your entire study.
Storage and Handling: Small Habits, Big Impact
Most peptide integrity issues come from unremarkable problems. A vial exposed to ambient conditions longer than planned. Repeatedly cycled in and out of controlled storage. Opened casually when the lab is busy. Over time, these small lapses accumulate into measurable drift.
For TB-500 peptide, the best storage habits are simple and realistic.
Keep exposure time short
When the vial is opened, treat it as focused work. Prepare what you need, seal it, and return it to controlled storage quickly. Avoid leaving the vial open while you handle unrelated tasks—those minutes matter.
Avoid repeated temperature cycling
Repeated warm and cold cycles can increase degradation risk over time. If your workflow requires multiple uses, plan around minimizing how often the same container is warmed, opened, and returned. Many labs reduce cycling by preparing a controlled stock and working from smaller portions when appropriate for their internal SOP.
Standardize storage behavior across your team
This is where labs often struggle silently. Two researchers can both be careful, but if their habits differ, the compound experiences different conditions. Shared inventory demands shared habits—and shared habits protect shared outcomes.
Reconstitution Math: Where “Peptide Problems” Actually Start
In many labs, the biggest hidden variable isn’t the compound—it’s the concentration. Not because anyone is careless, but because documentation is often incomplete. Someone writes “reconstituted TB-500” without recording the volume. Someone else assumes the old standard. Now two experiments meant to match don’t match, and nobody knows why.
With TB-500 peptide, the solution is straightforward: choose one standard reconstitution volume for your project and use it every time. Then document it in a format that’s impossible to misread later.
A clean documentation line records the reconstitution volume and the resulting concentration together, every single time. When those two numbers always appear together, assumptions disappear.
For a shared standard on conversions and dilution calculations, the Peptide Calculator ensures everyone does the math the same way using the same method. The tool itself isn’t the point—consistency is.
Your Repeatable TB-500 Workflow
If you want clean outcomes, treat intake and prep as part of the experiment—not overhead to rush through.
Receive and log. Log arrival date, product name, and lot number the day the vial arrives. Save the COA in a shared location tied to that lot so any team member can retrieve it instantly.
Verify before first use. Match the COA lot number to the vial label. Confirm the analytical method is stated. Make sure the document meets your internal quality standards.
Store immediately and consistently. Move the vial into controlled storage quickly. Reduce bench time and temperature cycling. Ensure multiple team members follow the same access behavior.
Prepare using one standard. Choose one reconstitution volume for your project’s TB-500 peptide work and stick to it. If another project needs a different concentration, treat it as a separate preparation batch with clear, explicit labeling.
Track usage across experiments. Record which lot and which preparation batch was used for each run. If outcomes drift, you can immediately check whether the shift aligns with a lot change, a prep date change, or a storage access pattern.
When your workflow is consistent, TB-500 peptide becomes the stable input your research needs.
TB-500 in a Broader Peptide Research Program
Most labs don’t work in isolation with one compound. They maintain a focused inventory aligned to study design. In tissue-response and cellular activity programs, it’s common to see TB-500 peptide alongside other well-characterized research peptides.
Some teams pair TB-500 peptide work with separate studies involving BPC-157 Peptide, while others keep copper peptide research distinct with GHK-Cu 100mg. Different compounds, identical reliability rules: log the lot, verify the COA, store consistently, prepare consistently, track what was used.
For a centralized view that keeps purchasing and naming consistent across your inventory, the Peptides catalog helps keep everything organized in one place.
Quick Diagnostic: Before You Redesign Your Protocol
If results ever start feeling noisy, check these fundamentals first:
Was the reconstitution volume identical across all runs for TB-500 peptide?
Did the lot change without being recorded in experiment notes?
Did the vial experience more warm-cold cycling than usual?
Were concentrations recorded in consistent units across team members?
Did different researchers handle the vial with different bench-time habits?
Most labs find the root cause right here. Fixing intake and preparation discipline is almost always faster—and cheaper—than redesigning your experiment.

Wrapping Up: Stable Inputs, Clean Results
The biggest advantage you can give your research is a stable, traceable input. TB-500 peptide becomes dramatically easier to work with when the lot is documented, the COA is verified, storage is consistent, and preparation math is standardized across your team.
Start with TB-500 Peptide (Thymosin Beta-4), keep your inventory organized through Peptides, and standardize calculations with the Peptide Calculator. When your workflow stays consistent, your outcomes become easier to interpret and far easier to reproduce.
Research Use Disclaimer: TB-500 peptide is sold for laboratory research use only. It is not intended for human consumption, diagnostic purposes, or therapeutic applications. Researchers should follow all applicable institutional and regulatory guidelines.
Frequently Asked Questions
How should I store TB-500 peptide for research?
Store TB-500 in controlled cold storage with minimal bench exposure. Avoid repeated temperature cycling by planning your access, and always return the vial to storage immediately after use. Standardize these storage habits across your entire team for consistent results.
Why is lot tracking important for TB-500 research?
Lot tracking lets you compare runs cleanly over time. If outcomes shift, you can quickly determine whether the change aligns with a lot change, a prep change, or a storage access pattern—saving weeks of troubleshooting.
How do I prevent concentration errors with TB-500?
Choose one standard reconstitution volume for your project and document it clearly. Require that every prep log includes both volume and final concentration together. Using a shared Peptide Calculator reference keeps conversions consistent across all researchers.